What about the sugars in breast milk?
Something that nearly always comes up when we talk about babies naturally being in ketosis is the fact that breast milk contains sugars — as much as 40% [1].Some people have even argued with us that therefore babies are not in ketosis!
So babies are born ketogenic. Breatsfeeding is better than formula. We're not sure why, but we say so, so there. http://t.co/fy9pqF7SAE
— Signe Rousseau (@Dr_Rousseau) February 7, 2014
@Dr_Rousseau @Jonathan_Witt "Assumption" of babies being ketogenic is wrong. Breast milk is 40% carbs, not ketogenic.
— Albie Cilliers (@albie_cilliers) February 7, 2014
That objection is, of course, reasoning backwards — objecting to a fact because it doesn’t fit a theory. That healthy, breastfed babies live in a state of ketosis and use the ketogenic metabolism for energy and growth is not a hypothesis; it is an empirical fact. See our article on ketogenic babies for details.
However, the fact that babies are in ketosis even while consuming a diet relatively high in carbohydrates does pose a mystery that deserves investigation. In this article, we’re going to suggest one possible explanation for the mystery, but remember that this possible explanation is just a hypothesis, until someone does an experiment to test it.
In brief
We can’t conclude, just because breast milk has a relatively high proportion of carbohydrates, that babies are burning a lot of carbohydrates for fuel.
- Breast milk is full of components that are good for building brains. Infancy is a period of intense brain growth.
- The sugars in breast milk are mostly from lactose, with small amounts in the form of oligosaccharides. Both lactose and oligosaccharides are replete with components that are crucial building blocks of brains.
- In addition to providing materials for growing brains, other non-fuel functions of at least oligosaccharides include serving as prebiotics and fighting infection.
- Insofar as some parts of the milk are being used for other purposes, they can’t also be used as fuel.
Therefore, a plausible explanation for how babies are in ketosis while consuming a relatively high-carbohydrate food, is that those carbohydrates are not being used as fuel, but rather as building blocks for the brain, and to a lesser extent, feeding gut bacteria, and fighting infections.
Lactose
Most of the carbohydrate in breast milk is lactose, which is broken down by digestion into glucose and galactose. Galactose is an important component of some glycoproteins and glycolipids, including cerebrosides — glycolipids in the brain and nervous system. Cerebrosides made of galactose are a major component of brain tissue [2]. They are also such a large component of myelin that cerebroside synthesis has been used as a measure of myelination or remyelination [2].
It is therefore plausible that much of the galactose in breast milk is used for brain tissue and myelin synthesis [3]. In fact, glucose is itself also used for making glycolipids for brain tissue [4], [5], although ketone bodies seem to be preferred [6], [7].
Oligosaccharides
After lactose and fat, oligosaccharides are the largest component of breast milk [8]. Oligosaccharides are unique to human breast milk — other animals produce almost no oligosaccharides in their milk [9].
Oligosaccharides are thought not to function as fuel. Some have been shown to have a prebiotic role [10], [11]. Much of the oligosaccharides pass completely through the infant’s digestive tract, and probably have an immune system function [12], [13]. Oligosaccharides also contain sialic acid [14], an important component in the brain used for cell-to-cell interactions, neuronal outgrowth, modifying synaptic connectivity, and memory formation [15].
Bottom line
The main point to take from all this is that many of the components of breast milk that one might presume to be used as “calories” are actually being used for other things, especially to make brains with. That includes glucose, galactose, proteins, fats, and even ketone bodies.
This could explain the fact that infants are in mild ketosis while breastfed, even though breast milk has higher carbohydrates than would support a ketogenic metabolism in an adult.
References
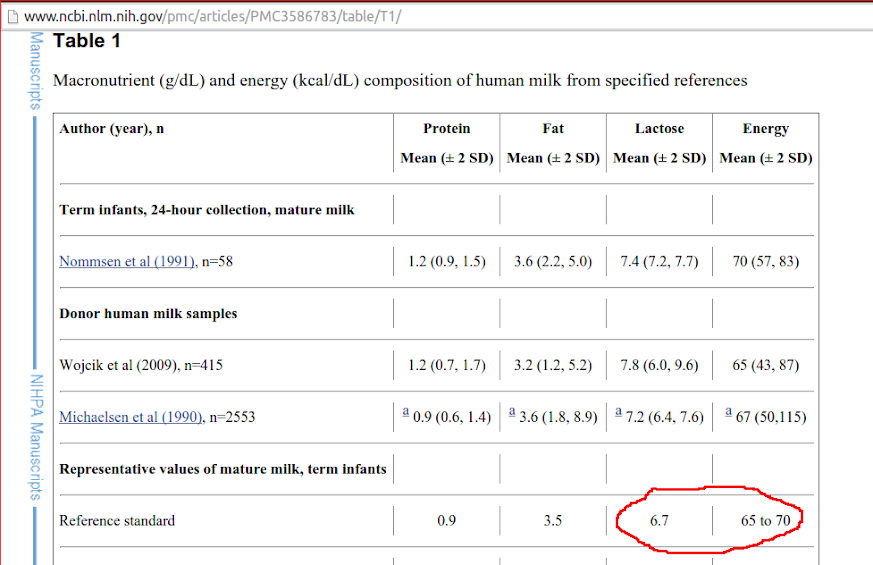
1. Calculating the macronutrients in breast milk is made very complex by not only the variation among individuals but diurnal variations, and variations over longer periods of time.
It is a huge simplification to report a single value for the amount of some component of breast milk:
Infant physiology, nutritional requirements, and lactational adequacy.
Whitehead RG.
Am J Clin Nutr. 1985 Feb;41(2 Suppl):447-58.
“It should be recognized, however, that we have all been guilty of adopting an oversimplified approach insofar as relating energy needs to milk volumes is concerned. The energy composition of milk is not the constant factor we have all tacitly assumed. Fat is the major energy-donating component and its concentrations vary considerably. At the beginning of each feed, from either breast, the fat content of the milk the baby receives is low, the exact level being determined by the extent to which that breast was emptied during the previous fed. As the baby feeds, fat content then rises by an amount that can be as much as 3-4-fold but the extent is very variable. There is also evidence that average fat levels vary at different times during the day in a cyclical manner. Even after one has taken account of these variables, it is still apparent that individual women have characteristically different fat concentrations in their breast milk. These complications have been extensively studied by Prentice in rural Gambian women (8, 9), and for the purpose of calculating breast milk requirements, they are almost impossible to untangle.”
Nonetheless, the standard reported amount of carbohydrate is 38―41%:
Human Milk Composition: Nutrients and Bioactive Factors
Olivia Ballard, JD, PhD (candidate) and Ardythe L. Morrow, PhD, MSc
Pediatr Clin North Am. Feb 2013; 60(1): 49–74.
“Galactosylceramide is the principal glycosphingolipid in brain tissue. Galactosylceramides are present in all nervous tissues, and can compose up to 2% dry weight of grey matter and 12% of white matter. They are major constituents of oligodendrocytes.”
“Monogalactosylceramide is the largest single component of the myelin sheath of nerves. Cerebroside synthesis can therefore give a measurement of myelin formation or remyelination.”
3. I first heard this idea from this blog post:
What can we learn from breast milk? Part 1: Macronutrients
“…the carbohydrate source is lactose, made of glucose and galactose. Now galactose is very special, it’s not used as an energy fuel like glucose, it’s used for myelin synthesis (that is making nerve insulation), this is why human breast milk is so high in lactose, for the galactose! So that ~15% becomes ~7% of calories coming from carbs for an adult (~38g @ 2000 calories).”
4. Evidence type: review
Energy metabolism in developing brain cells.
Edmond J.
Can J Physiol Pharmacol. 1992;70 Suppl:S118-29.
“Many studies in the decade, 1970-1980, in human infants and in the rat pup model show that both glucose and the ketone bodies, acetoacetate and D-(-)-3-hydroxybutyrate, are taken up by brain and used for energy production and as carbon sources for lipogenesis. Products of fat metabolism, free fatty acids, ketone bodies, and glycerol dominate metabolic pools in early development as a consequence of the milk diet. This recognition of a distinctive metabolic environment from the well-fed adult was taken into consideration within the last decade when methods became available to obtain and study each of the major cell populations, neurons, astrocytes, and oligodendrocytes in near homogeneous state in primary cultures. Studies on these cells made it possible to examine the distinctive metabolic properties and capabilities of each cell population to oxidize the metabolites that are available in development. Studies by many investigators on these cell populations show that all three can use glucose and the ketone bodies in respiration and for lipogenesis.”
5. Evidence type: non-human animal experiment
Biosynthesis of phospholipids and sphingolipids from acetoacetate and glucose in different regions of developing brain in vivo.
Yeh YY.
J Neurosci Res. 1984;11(4):383-94.
“The incorporation of 14C-label from subcutaneously injected [3-14C]acetoacetate and [U-14C]glucose into phospholipids and sphingolipids in different regions of developing rat brain was determined. In all regions, phosphatidylcholine was the lipid synthesized most readily from either substrate. The percentages of radioactivity in other phospholipids and most sphingolipids remained relatively constant throughout postnatal development. An exceptional increase in the percentage of radioactivity incorporated into cerebroside, coinciding with a decrease of incorporation into phosphatidylcholine, was first noted on day 12 and continued until a maximal level was reached between days 18 and 20 of postnatal age. These developmental changes in preferential synthesis of lipids were associated with increased demands for phospholipids and cerebroside during the early and late postnatal stages, respectively. There was no difference in accumulation of radioactivity from acetoacetate, expressed as dpm of [14C]acetoacetate recovered in phospholipids plus sphingolipids per g of tissue, among all brain regions during the first 5 days of life. During active myelination (12 to 20 days of age); however, the amount of 14C-label was highest in brain stem, ranging from 1.9- to 2.3-fold greater than values for cerebrum and thalamus. The region with the next highest accumulation was cerebellum, followed by midbrain. During the same period, brain stem was likewise the most active site of accumulation of radioactivity from 14C-labeled glucose. Higher amounts of [14C]acetoacetate label accumulated in lipids of brain stem and cerebellum, relative to midbrain, thalamus, and cerebrum, coincide with evidence that active myelination begins in the hindbrain and proceeds rostrally toward the forebrain. Ketone bodies could therefore serve as a potential source of phospholipids and sphingolipids for brain growth and maturation.”
6. Evidence type: non-human animal experiment
Preferential utilization of ketone bodies in the brain and lung of newborn rats.
Yeh YY, Sheehan PM.
Fed Proc. 1985 Apr;44(7):2352-8.
(Emphasis ours)
“Persistent mild hyperketonemia is a common finding in neonatal rats and human newborns, but the physiological significance of elevated plasma ketone concentrations remains poorly understood. Recent advances in ketone metabolism clearly indicate that these compounds serve as an indispensable source of energy for extrahepatic tissues, especially the brain and lung of developing rats. Another important function of ketone bodies is to provide acetoacetyl-CoA and acetyl-CoA for synthesis of cholesterol, fatty acids, and complex lipids. During the early postnatal period, acetoacetate (AcAc) and beta-hydroxybutyrate are preferred over glucose as substrates for synthesis of phospholipids and sphingolipids in accord with requirements for brain growth and myelination. Thus, during the first 2 wk of postnatal development, when the accumulation of cholesterol and phospholipids accelerates, the proportion of ketone bodies incorporated into these lipids increases. On the other hand, an increased proportion of ketone bodies is utilized for cerebroside synthesis during the period of active myelination. In the lung, AcAc serves better than glucose as a precursor for the synthesis of lung phospholipids. The synthesized lipids, particularly dipalmityl phosphatidylcholine, are incorporated into surfactant, and thus have a potential role in supplying adequate surfactant lipids to maintain lung function during the early days of life. Our studies further demonstrate that ketone bodies and glucose could play complementary roles in the synthesis of lung lipids by providing fatty acid and glycerol moieties of phospholipids, respectively. The preferential selection of AcAc for lipid synthesis in brain, as well as lung, stems in part from the active cytoplasmic pathway for generation of acetyl-CoA and acetoacetyl-CoA from the ketone via the actions of cytoplasmic acetoacetyl-CoA synthetase and thiolase.”
7. Evidence type: non-human animal experiment
Ketone body metabolism in the neonate: development and the effect of diet.
Edmond J, Auestad N, Robbins RA, Bergstrom JD.
Fed Proc. 1985 Apr;44(7):2359-64.
(Emphasis ours)
“In the course of mammalian development milk has evolved with unique characteristics as has the capacity of the neonatal rat to process this nutrient source. The primary carbon source in milk is fat, which provides two readily utilized metabolites, acetoacetate and D(-)-3 hydroxybutyrate (ketone bodies), as well as free fatty acids and glycerol. Carbohydrate provides less than 12% of the caloric content of rat milk and glucose has to be produced by the suckling rat to maintain glucose homeostasis. One would predict that glucose would be used sparingly and in pathways that cannot be satisfied by other readily available metabolites. Studies of the uptake of metabolites and the development of key enzymes for the utilization of glucose and ketone bodies by developing brain support the concept that ketone bodies are preferred substrates for the supply of carbon to respiration and lipogenesis. Astrocytes, oligodendrocytes, and neurons from developing brain all have an excellent capacity to use ketone bodies for respiration. By contrast, glucose is utilized preferentially in the hexose monophosphate shunt by all three cell populations. We are examining the requirement for ketone bodies by developing brain with the application of a system to rear rat pups artificially on a milk substitute that promotes a hypoketonemia.”
8. Evidence type: review
[Human milk oligosaccharides. The rule in the health and development of the infants]. [Article in Spanish]
Gudiel-Urbano M1, Goñi I.
Arch Latinoam Nutr. 2001 Dec;51(4):332-9.
(Emphasis ours)
“Breast-feeding is the optimal mode of feeding for the normal full-term infant. Human milk composition knowledge has been basis for recommended dietary allowances for infants. Few studies about human milk carbohydrates have been done until the last decade. However, carbohydrates provide approximately 40-50% of the total energy content of breast milk. Quantitatively oligosaccharides are the third largest solute in human milk after lactose and fat. Each individual oligosaccharide is based on a variable combination of glucose, galactose, sialic acid, fucose and N-acetylglucosamine with many and varied linkages between them, thus accounting for the enormous number of different oligosaccharides in human milk. The oligosaccharides content in human milk varies with the duration of lactation, diurnally and with the genetic makeup of the mother. At present, a great interest in the roles of human milk oligosaccharides is raising. They act as a the soluble fibre in breast milk and their structure is available to act as competitive ligands protecting the breast-fed infant from pathogens and act as well as prebiotic. They may also act as source of sialic acid and galactose, essential for brain development. This is why today there is an increasing health and industrial interest in human milk oligosaccharides content, with the main purpose of incorporating them as new ingredients in infant nutrition.”
9. Evidence type: review
Human milk oligosaccharides: only the breast.
McVeagh P1, Miller JB.
J Paediatr Child Health. 1997 Aug;33(4):281-6.
“Abstract
“Over 100 years ago it was first deduced that a major component of human milk must be an unidentified carbohydrate that was not found in cows milk. At first this was thought to be a form of lactose and was called gynolactose. We now know that this was not a single carbohydrate but a complex mixture of approximately 130 different oligosaccharides. Although small amounts of a few oligosaccharides have been found in the milk of other mammals, this rich diversity of sugars is unique to human milk. The oligosaccharide content of human milk varies with the infant’s gestation, the duration of lactation, diurnally and with the genetic makeup of the mother. Milk oligosaccharides have a number of functions that may protect the health of the breast fed infant. As they are not digested in the small intestine, they form the ‘soluble’ fibre of breast milk and their intact structure is available to act as competitive ligands protecting the breast-fed infant from pathogens. There is a growing list of pathogens for which a specific oligosaccharide ligand has been described in human milk. They are likely to form the model for future therapeutic and prophylactic anti-microbials. They provide substrates for bacteria in the infant colon and thereby contribute to the difference in faecal pH and faecal flora between breast and formula-fed infants. They may also be important as a source of sialic acid, essential for brain development.”
10. Evidence type: review
The first prebiotics in humans: human milk oligosaccharides.
Coppa GV, Bruni S, Morelli L, Soldi S, Gabrielli O.
J Clin Gastroenterol. 2004 Jul;38(6 Suppl):S80-3.
“The development of intestinal microflora in newborns is strictly related to the kind of feeding. Breast-fed infants, unlike the bottle-fed ones, have an intestinal ecosystem characterized by a strong prevalence of bifidobacteria and lactobacilli. Data available so far in the literature show that, among the numerous substances present in human milk, oligosaccharides have a clear prebiotic effect. They are quantitatively one of the main components of human milk and are only partially digested in the small intestine, so they reach the colon, where they stimulate selectively the development of bifidogenic flora. Such results have been recently proved both by characterization of oligosaccharides in breast-fed infant feces and by the study of intestinal microflora using new techniques of molecular analysis, confirming that human milk oligosaccharides represent the first prebiotics in humans.”
11. Evidence type: review
Prebiotics in human milk: a review.
Coppa GV, Zampini L, Galeazzi T, Gabrielli O.
Dig Liver Dis. 2006 Dec;38 Suppl 2:S291-4.
“The microbic colonization of human intestine begins at birth, when from a sterile state the newborn is exposed to an external environment rich in various bacterial species. The kind of delivery has an important influence on the composition of the intestinal flora in the first days of life. Thereafter, the microflora is mainly influenced by the kind of feeding: breast-fed infants show a predominance of bifidobacteria and lactobacilli, whereas bottle-fed infants develop a mixed flora with a lower number of bifidobacteria. The “bifidogenic effect” of human milk is not related to a single growth-promoting substance, but rather to a complex of interacting factors. In particular the prebiotic effect has been ascribed to the low concentration of proteins and phosphates, the presence of lactoferrin, lactose, nucleotides and oligosaccharides. The real prebiotic role of each of these substances is not yet clearly defined, with the exception of oligosaccharides which undoubtedly promote a bifidobacteria-dominant microflora.”
12. Evidence type: review
Human milk oligosaccharides: only the breast.
McVeagh P, Miller JB.
J Paediatr Child Health. 1997 Aug;33(4):281-6.
(Emphasis ours)
“Over 100 years ago it was first deduced that a major component of human milk must be an unidentified carbohydrate that was not found in cows milk. At first this was thought to be a form of lactose and was called gynolactose. We now know that this was not a single carbohydrate but a complex mixture of approximately 130 different oligosaccharides. Although small amounts of a few oligosaccharides have been found in the milk of other mammals, this rich diversity of sugars is unique to human milk. The oligosaccharide content of human milk varies with the infant’s gestation, the duration of lactation, diurnally and with the genetic makeup of the mother. Milk oligosaccharides have a number of functions that may protect the health of the breast fed infant. As they are not digested in the small intestine, they form the ‘soluble’ fibre of breast milk and their intact structure is available to act as competitive ligands protecting the breast fed infant from pathogens. There is a growing list of pathogens for which a specific oligosaccharide ligand has been described in human milk. They are likely to form the model for future therapeutic and prophylactic anti-microbials. They provide substrates for bacteria in the infant colon and thereby contribute to the difference in faecal pH and faecal flora between breast and formula-fed infants. They may also be important as a source of sialic acid, essential for brain development.”
13. Evidence type: experiment
Survival of human milk oligosaccharides in the intestine of infants.
Chaturvedi P, Warren CD, Buescher CR, Pickering LK, Newburg DS.
Adv Exp Med Biol. 2001;501:315-23.
(Emphasis ours)
“Several human milk oligosaccharides inhibit human pathogens in vitro and in animal models. In an infant, the ability of these oligosaccharides to offer protection from enteric pathogens would require that they withstand structural modification as they pass through the alimentary canal or are absorbed and excreted in urine. We investigated the fate of human milk oligosaccharides during transit through the alimentary canal by determining the degree to which breast-fed infants’ urine and fecal oligosaccharides resembled those of their mothers’ milk. Oligosaccharide profiles of milk from 16 breast-feeding mothers were compared with profiles of stool and urine from their infants. Results were compared with endogenous oligosaccharide profiles obtained from the urine and feces of age-, parity-, and gender-matched formula-fed infants. […] Among breast-fed infants, concentrations of oligosaccharides were higher in feces than in mothers’ milk, and much higher in feces than in urine. Urinary and fecal oligosaccharides from breast-fed infants resembled those in their mothers’ milk. Those from formula-fed infants did not resemble human milk oligosaccharides, were found at much lower concentrations, and probably resulted from remodeling of intestinal glycoconjugates or from intestinal bacteria. Most of the human milk oligosaccharides survived transit through the gut, and some were absorbed and then excreted into the urine intact, implying that inhibition of intestinal and urinary pathogens by human milk oligosaccharides is quite likely in breast-fed infants.”
14. Evidence type: experiment
Sialic acid in human milk: composition and functions.
Nakano T1, Sugawara M, Kawakami H.
Acta Paediatr Taiwan. 2001 Jan-Feb;42(1):11-7.
“Breast milk is the best nutrient source for infants. It contains all elements needed for a normal growth and development of infants. Human milk contains a large amount of sialic acid compared with bovine milk. Sialic acid contained in oligosaccharides, glycolipids and glycoproteins in milk is considered to play important roles in physiological functions in infancy. Thus, we have investigated the sialic acid composition and the functions of sialylated compounds in human milk. Sialic acids comprise a family of neuraminic acid derivatives present in secretions, fluids and tissues of mammals. In milk, sialic acid is present in different sialoglycoconjugate compounds such as oligosaccharides, glycolipids and glycoproteins, not in a free form. Human milk contains 0.3-1.5 mg/ml of sialic acid. Sialic acid bound to oligosaccharides accounts for about 75% of the total sialic acid contained in human milk. Most of the sialic acid contained in human milk is found in the form of sialyllactose, an oligosaccharide formed from lactose and sialic acid. In milk, gangliosides, sialic acid-containing glycolipid, occur mainly as monosialoganglioside 3 (GM3) and disialoganglioside 3 (GD3). The concentration of GM3 in human milk increases, while that of GD3 concentration decreases during lactation. Because the brain and central nervous system contain considerable level of sialic acid in infancy, it is considered to play important roles on the expression and development of their functions. Moreover, we found that some sialylated compounds had inhibited the adhesion of toxins, bacteria and viruses to the receptors on the surface of epithelial cells. Additionally, we found that some sialylated compounds had growth-promoting effects on bifidobacteria and lactobacilli, predominantly present in the intestinal flora of infants fed with human milk. The results suggested that sialylated compounds in human milk possibly behaved as a physiological component in the intestinal tract of infants to protect them against enteric infections.”
15. Evidence type: review
Sialic acid is an essential nutrient for brain development and cognition.
Wang B.
Annu Rev Nutr. 2009;29:177-222. doi: 10.1146/annurev.nutr.28.061807.155515.
“The rapid growth of infant brains places an exceptionally high demand on the supply of nutrients from the diet, particularly for preterm infants. Sialic acid (Sia) is an essential component of brain gangliosides and the polysialic acid (polySia) chains that modify neural cell adhesion molecules (NCAM). Sia levels are high in human breast milk, predominately as N-acetylneuraminic acid (Neu5Ac). In contrast, infant formulas contain a low level of Sia consisting of both Neu5Ac and N-glycolylneuraminic acid (Neu5Gc). Neu5Gc is implicated in some human inflammatory diseases. Brain gangliosides and polysialylated NCAM play crucial roles in cell-to-cell interactions, neuronal outgrowth, modifying synaptic connectivity, and memory formation. In piglets, a diet rich in Sia increases the level of brain Sia and the expression of two learning-related genes and enhances learning and memory. The purpose of this review is to summarize the evidence showing the importance of dietary Sia as an essential nutrient for brain development and cognition.”
